A PARACHUTE for Ischemic Heart Failure
January 26, 2017
Harrington Heart & Vascular Institute Innovations – Winter 2017 – View Full PDF
 Hiram Bezerra, MD, PHD
Hiram Bezerra, MD, PHDDamaged cardiac muscle after a heart attack can lead to heart enlargement and decline in ventricular performance. This may result in fatigue and shortness of breath, severely impacting quality of life. In these cases, studies suggest that an alternative implantable device can improve symptoms and quality of life.
The Percutaneous Ventricular Restoration Device (Parachute®), made by CardioKinetix, is the first minimally invasive treatment for such patients. This novel ventricular partitioning device (VPD) is implanted with a catheter inserted via the femoral artery. University Hospitals Harrington Heart & Vascular Institute is leading worldwide clinical investigation of this device, exploring its value for patients with reduced blood supply to the heart (ischemic heart failure).
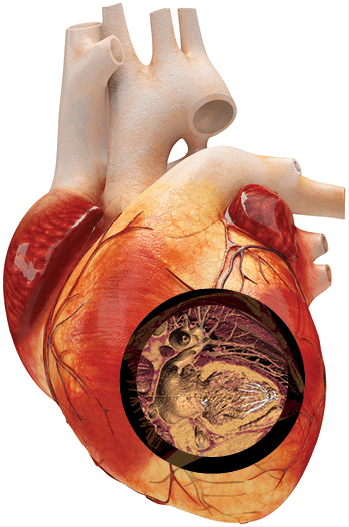
In a 2016 study published in Catheterization and Cardiovascular Interventions, a team of UH researchers assessed the safety, feasibility and primary efficacy of VPD and found that the quality of implantation impacted clinical outcomes. The team concluded that implanting the Parachute device in the proper position is essential for good outcomes. The foot of the device must sit at the left ventricle (LV) apex with the nitinol struts anchored to the LV wall.
“Although still under FDA investigation, Parachute has emerged as a safe and feasible treatment option,” says Hiram Bezerra, MD, PhD, the study’s senior author, Director, Cardiac Catheterization Laboratories, University Hospitals Cleveland Medical Center; Associate Professor of Medicine, Case Western Reserve University School of Medicine. “It is the only percutaneous, nonsurgical mechanical intervention for heart failure.”
Parachute separates damaged heart muscle from healthy muscle, and cardiac output and function of the LV can be restored. Patients remain awake during the procedure, which typically takes just 30 minutes in a cardiac catheterization lab and does not involve the risks associated with surgical alternatives. Early research results show the Parachute device may minimize hospital readmissions due to complications from heart failure.
Implanting the device requires a cardiac computed tomography (CCT) scan in advance. UH’s Cardiovascular Imaging Core Laboratory receives all CCT images worldwide from the current clinical trial in the U.S. and commercial cases in Europe and Asia. UH researchers then recommend the size of Parachute device to be implanted. Marco Costa, MD, PhD, President of UH Harrington Heart & Vascular Institute, is the study’s principal investigator.
“We’re in a unique position of expertise by reviewing all CCT scans globally from this study,” Dr. Bezerra says. “Each case passes through us before any of the devices are implanted. We can apply this great expertise to our own patients.”
For more information or to refer a patient, call 216-844-3800 or email HVInnovations@UHhospitals.org.
Tags: Heart Failure


