Contrast Enhanced Ultrasound Imaging Holds Promise for Prostate Cancer Detection Treatment
November 16, 2021
Innovations in Urology | Fall 2021
Urologists predominantly use systematic biopsies of areas of interest to diagnose prostate cancer. While contrast agents may be used with ultrasound to guide these biopsies, they are not as sensitive and specific as clinicians would like. Three of University Hospitals' and Case Western Reserve University School of Medicine researchers are trying to build a better mousetrap to improve the accuracy and early detection of prostate cancer.
 Agata A. Exner, PhD
Agata A. Exner, PhD“While contrast agents have been widely used in the international ultrasound community, they’re not as widely used in the U.S.,” says Agata Exner, PhD, Director of the Case Center for Imaging Research at Case Western Reserve University School of Medicine. “We’ve developed what we believe is a better ultrasound contrast agent.”
Nanotech overcomes limitations in microbubbles
Microbubbles are clinical agents used in ultrasound technology. However, they are about the size of a red blood cell, which limits them to blood vessels only. Furthermore, they only last about five minutes, reducing their clinical usefulness.
“In contrast, nanoparticles are very small, highly deformable and have low density,” Exner says, “so they can extravasate blood vessels and target specific cells, in this case, prostate cancer cells. They also last longer in circulation.”
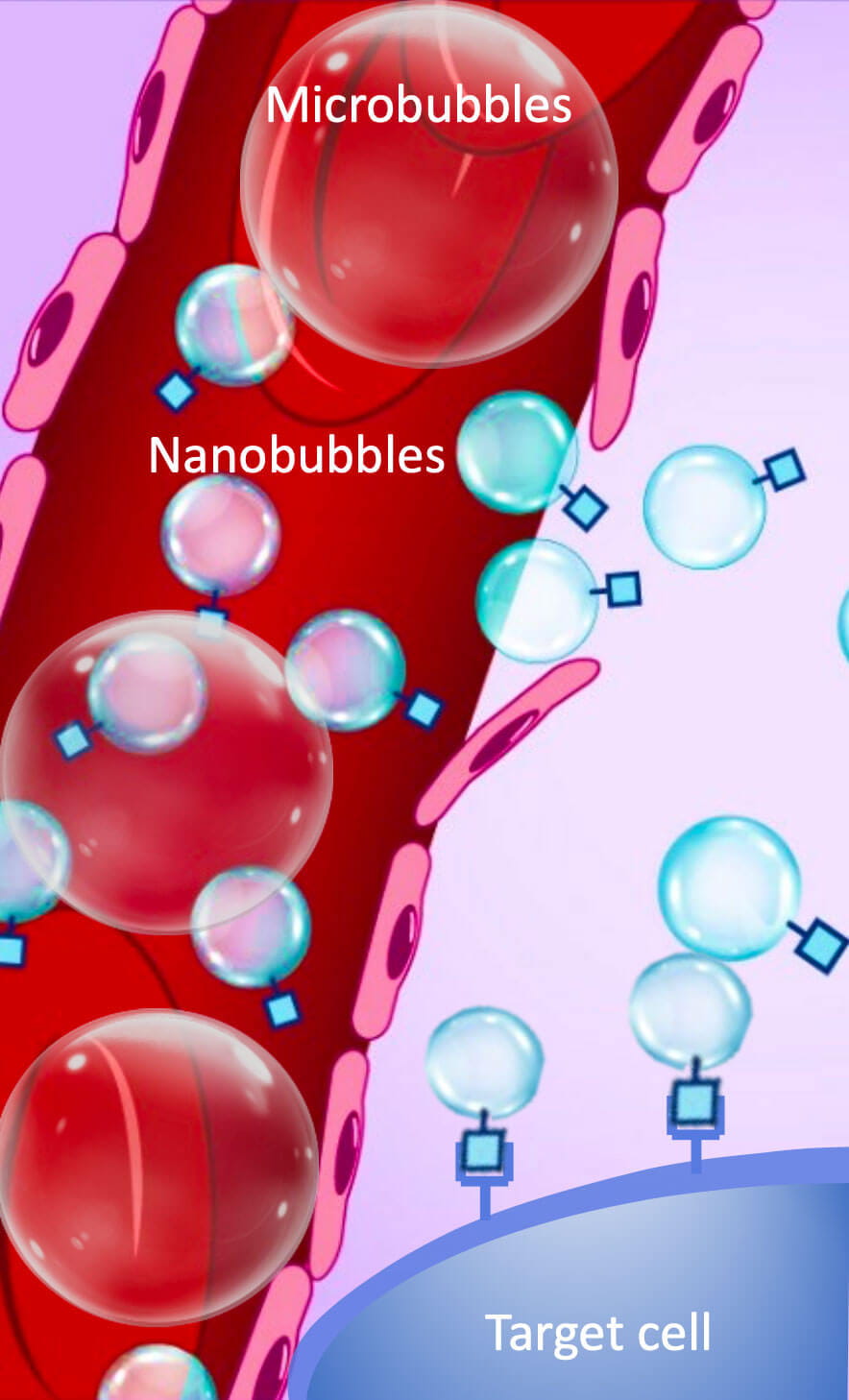 This schematic is adapted from original work created by Erika Woodrum, Certified Medical Illustrator. Science Highlights 5/5/21
This schematic is adapted from original work created by Erika Woodrum, Certified Medical Illustrator. Science Highlights 5/5/21Exner — in collaboration with School of Medicine professors James Basilion, PhD, and Lee Ponsky, MD, who is also Chair of the Department of Urology at UH Cleveland Medical Center — have developed new materials for imaging purposes using nanoparticles that can serve as contrast agents for ultrasound imaging.
“A nanobubble is a lipid particle that is about 300 nanometers, 300 times smaller than the width of a human hair,” Exner says. “The outside is a lipid and the inside is filled with gas. When you image a bubble in an acoustic field of ultrasound, the lipid shell vibrates and the gas reflects sound waves in a very different way than the rest of the body. It enables us to visualize the area of interest with very high sensitivity of detection.”
Using nanobubbles to improve prostate cancer detection
These nanobubbles target tumors that express prostate specific membrane antigen (PSMA), which has been correlated to aggressiveness of prostate cancer. As a molecular biologist, Basilon has also developed a better ligand to target PSMA.
“Combining the better ligand with nanobubble technology, we can now detect and delineate prostate tumors within the prostate gland,” Exner says. “This technology will not change a urologist’s workflow, but introduces the administration of a contrast agent that helps target biopsies to a region of interest in real time during a transrectal ultrasound.”
The benefits to urologists are significant, Exner says. Currently, the transrectal biopsy with ultrasound only shows the location of the prostate gland. It’s difficult to see if there is a tumor in the gland itself.
Nanobubbles with a contrast agent have much higher efficiency in the biopsy process and eliminate false positives and false negatives, providing more effective detection of prostate cancer. They also help urologists detect lesions in the first biopsy, hopefully eliminating the need for subsequent biopsies, which reduces patient morbidity.
Down the road, Exner says, urologists may be able to use the contrast ultrasound scan to exclude the possibility someone has cancer, avoiding a biopsy altogether.
This research is in the preclinical stage, she emphasizes. The next step is to gather enough information to file an investigational new drug (IND) application with the U.S. Food and Drug Administration.
“Once the IND process is approved, we’ll be able to manufacture the bubbles in scaled up fashion and then begin clinical trials with humans,” Exner says.
The future of nanobubbles
Exner is excited about some of the other potential uses of nanobubbles. Because the bubbles are fragile, they can be popped with ultrasound at a specific location.
“We’re thinking of potential theranostic applications, where we can visualize treatment and use the particles to carry drugs to the prostate cancer cells. This would minimize side effects as the stress of any toxic drug would be limited to the prostate cancer cells themselves.”
In addition to potentially carrying antineoplastic drugs, there may be opportunities for other therapeutic uses. Results of Exner’s preliminary studies of nanobubbles in prostate cancer treatment — conducted in collaboration with Dr. Michael Kolios of Ryerson University and Dr. Greg Czarnota of Sunnybrook Research Institute in Toronto — showed that nanobubble-radiation combined treatment resulted in twice as much cellular death compared to just nanobubble treatment alone.
Prostate cancer is the second-deadliest cancer in men in the U.S. and a leading cause of cancer-related deaths. The National Cancer Institute estimates that almost 250,000 men will be diagnosed with prostate cancer in 2021 (13 percent of all new cancers) and 34,000 will die. Any tool that improves the sensitivity and specificity of tumor detection would be a welcome addition to urologists’ clinical toolbox.
For more information about nanobubble technology, email Agata Exner at agata.exner@case.edu.
Reference: Building better bubbles for ultrasound could enhance image quality, facilitate treatments. Science Highlights: May 25, 2021


