
Lorraine and Bill Dodero Limb Preservation Center
A specialized team at University Hospitals Harrington Heart & Vascular Institute is transforming the standard of care for “no option” critical limb ischemia (CLI) patients at risk of amputation. If you or a loved one have been told that amputation is your only treatment option, it is vital that you get a second opinion from our experts. We have the expertise to save limbs with advanced, innovative techniques – even when other treatment options have failed.
Make an Appointment
Call 216-286-9117 to schedule an appointment.
Find an amputation prevention specialistAvoid Amputation with Unique Expertise
Study Shows Significantly Lower Amputation Rates
UH Harrington Heart & Vascular Institute led a first-of-its-kind study with results published in Circulation: Cardiovascular Interventions showing that 75 percent of patients scheduled for a major amputation were instead able to keep their limb after being managed by our Limb Salvage Advisory Council.
Read MoreUniversity Hospitals Harrington Heart & Vascular Institute’s Lorraine and Bill Dodero Limb Preservation Center is one of just a few that exist in the United States, providing limb preservation and lifesaving results for patients with blood flow blockages in the legs.
We have assembled a unique, interdisciplinary team to evaluate alternative treatments for patients scheduled for amputation. Our Limb Salvage Advisory Council (LSAC) is comprised of vascular surgeons, endovascular interventionalists, vascular medicine specialists, podiatrists and wound care experts – including multiple physicians from each specialty to ensure that different points of view are considered. This professional collaboration often leads to more potential treatment options to avoid amputation.
Who Can Benefit from an Amputation Prevention Consultation?
A Lifesaving Option for Patients Facing Limb Amputation
Mehdi Shishehbor, DO, MPH, PhD, President, University Hospitals Harrington Heart & Vascular Institute, explains how the Lorraine and Bill Dodero Limb Preservation Center is saving lives and limbs. “Don’t accept amputation as an answer,” he says. “Get a second and third opinion.”
Watch NowDiabetes-related circulation issues in the legs or peripheral artery disease (PAD) can cause cramping, numbness and nonhealing wounds that can affect a person’s daily activities. If left untreated or poorly managed, blood flow blockages in the legs can lead to:
If treatments such as angioplasty and stent placement, open bypass, thrombectomy or other endovascular repairs don’t help, patients may be told amputation is the recommended course of action. However, the Lorraine and Bill Dodero Limb Preservation Center offers potential solutions that other institutions don’t.
Unparalleled Access to the Best Limb Salvage Therapies
$5 Million Gift from Lorraine and Bill Dodero Creates One-of-a-Kind Limb Preservation Center
The Lorraine and Bill Dodero Limb Preservation Center offers the most advanced therapies available to reduce leg amputations, giving our patients the best options to prevent lower extremity limb loss. These catheter-based procedures provide a minimally invasive treatment option, even for those deemed too sick or high risk for open surgery. Available therapies and procedures include:
- LimFlow Percutaneous Deep Vein Arterialization (pDVA) System
-
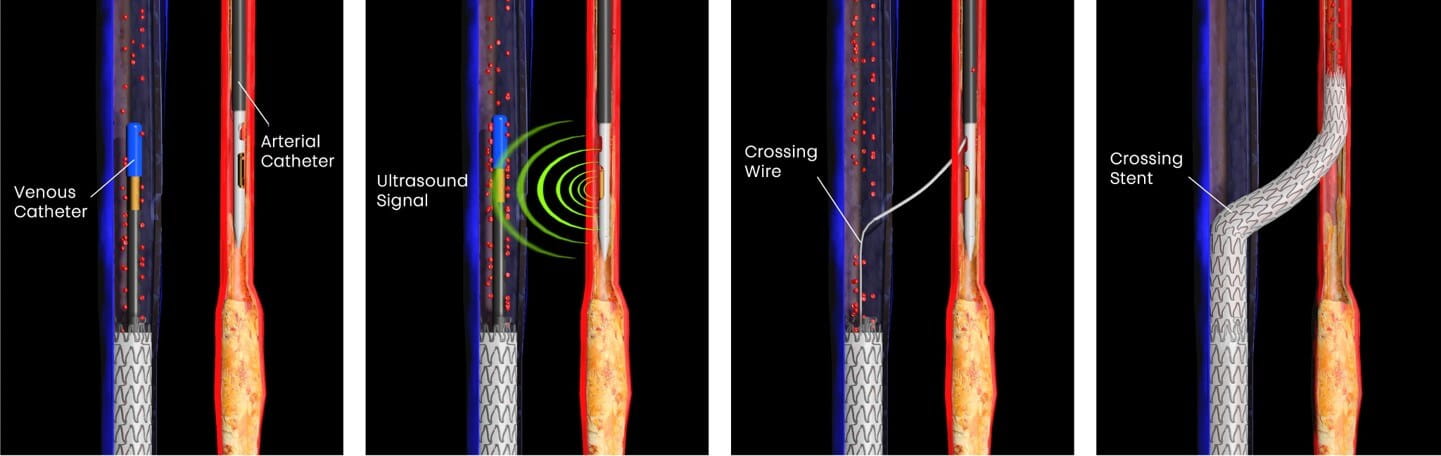
This breakthrough FDA-approved procedure employs a novel proprietary technique to provide endovascular venous arterialization that cannot be corrected by surgery or other endovascular approaches. The goal is to redirect blood from ischemic arteries into the tibial veins, providing a bypass around the blockage in the artery and resupplying oxygen to the surrounding tissue. Outcomes from the procedure include wound healing, limb salvage and remarkable changes in the vasculature of the foot.
- Biologic Therapy
-
Used in conjunction with revascularization, biological therapy is injected into the foot to observe wound healing in a more timely fashion. The biologic stimulates and directs naturally occurring angiogenic factors (formation of new blood vessels) in the body to the site of injury with the hope that new small blood vessels will build to improve circulation.
- Resorbable Stents
-
Made of materials like those used in dissolving sutures, these stents are not a permanent implant, but are reabsorbed over two years, leaving the artery healthy and without any metal inside the body.
- Drug-Coated Balloons
-
This novel device consists of a balloon coated in medicine. Once the balloon is in place, the medication is released slowly over time, allowing it to reach the inner layer of the arteries being treated. This delivery method minimizes the amount of medication lost in the bloodstream and improves overall wound healing.
- Radial to Peripheral (R2P) Procedures
-
R2P allows complex procedures in the leg to be performed through the wrist. The last five to 10 years have shown when patients are treated from the wrist, it is less risky and associated with less bleeding, enhanced patient satisfaction and a higher quality procedure than if done through the groin.
International Training Site for Optimal Limb Salvage Techniques
Center for Novel and Integrated Approaches in Vascular-Metabolic Disease
One of a select few programs in the United States focused on the interdisciplinary treatment of patients with type 2 diabetes and peripheral artery disease (PAD) or critical limb-threatening ischemia (CLTI).
Learn MoreOur experts are committed to education and public health. We have trained more than 400 physicians across the globe in advanced techniques for limb salvage and are highly sought as guest lecturers at medical conferences. We remain at the forefront of innovation in limb salvage and amputation prevention with our work continuously published in peer-reviewed medical journals. Examples of recent publications include:
- UH Harrington Leads Pivotal Study of LimFlow Therapy for Chronic Limb Threatening Ischemia
- Treatment Below the Knee Disease in Chronic Limb Threatening Ischemia
- UH Team Treats Case of No Option CLTI with Off the Shelf Percutaneous Deep Vein Arterialization
Patient Success Stories
With a steadfast mantra of “leave no limbs behind,” we are dedicated to delivering the best care. Learn about patients who were at risk for amputation because of advanced vascular disease but found hope – and ultimately saved their limbs – with help from the team at University Hospitals Harrington Heart & Vascular Institute.
UH Team Saves Leg and Life of Kentucky Man Facing Amputation

After three different doctors told Daniel Boynton his leg needed to be amputated, he traveled to Cleveland for a limb- and life-saving procedure.
Novel Council at UH Harrington Heart & Vascular Institute Diverts Patients from Amputation

Maurice Edwards of Cleveland knew he might lose his leg to amputation, but thanks to a novel council at University Hospitals (UH) Harrington Heart & Vascular Institute, he was spared that fate.
Leg Saved with Advanced Procedure
With one leg previously amputated, Jean came to UH with the same condition – critical limb ischemia (CLI) – in her other leg. Through advanced techniques, her leg was saved.
Doctors Save Leg of Patient With Diabetes By Restoring Blood Flow

John underwent three failed procedures to restore circulation to his foot. Doctors told him his leg would need to be amputated, so John sought a second opinion.
Limb Preservation Doctors
Mehdi Shishehbor, DO, MPH, PhD
- Cardiology-Interventional Cardiology, Cardiology, Cardiology-Vascular Medicine
-
Cleveland (0 mi.)
Cleveland (0 mi.)
Beachwood (7 mi.)
Beachwood (7 mi.)
Vascular Medicine Specialists
Our vascular medicine specialists focus on the diagnosis and medical management of any circulatory condition affecting the blood vessels, including arteries, veins and lymphatic system. They may also refer patients to other members of the Vascular Center, including an interventional cardiologist or vascular surgeon for advanced therapies if needed.
Vascular Surgeons
Our vascular surgeons are dedicated to the evaluation and treatment of any vascular condition outside the heart or brain. They are trained to perform every possible vascular intervention and surgery, including the most advanced procedures when other treatments are not effective or appropriate based on the patient’s condition.
Endovascular Specialists
Our endovascular medicine specialists in the Vascular Center are interventional cardiologists who have completed additional, focused training in the management of patients with vascular disease as experts in performing minimally invasive, catheter-based procedures to restore blood flow, reduce pain or assist with wound healing.
Vascular Advanced Practice Providers
Our specially trained physician assistants and nurse practitioners in the Vascular Center work in close collaboration with physician team members. They meet with patients to review records, perform physical exams, order related testing and provide education about the patient’s vascular condition or procedure.
Lorraine and Bill Dodero Limb Preservation Center
If you or a loved one have been told that amputation is your only treatment option, it is vital that you get a second opinion from our experts. We have the expertise to save limbs with advanced, innovative techniques – even when other treatment options have failed.
Call 216-286-9117 to schedule an appointment.



