Research
Our laboratory has a keen focus on congenital heart defects and understanding their underlying mechanisms towards therapeutic intervention.
We also investigate other areas of connective tissue biology including aortic development and disease as well as musculoskeletal development and disease. The main goals of these projects are to determine if changes in the extracellular matrix will alter development and disease and learn how to correct them to enhance patient care.
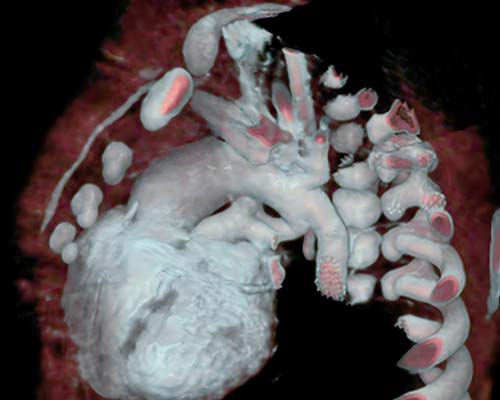
Congenital heart disease is the most common birth defect and affect approximately 1% of births per year in the United States. We genetically engineered preclinical mouse and cell culture models with patient gene mutations to identify the function of the mutated gene that is causative in congenital heart defects. We partner with geneticists and employ genomics and proteomics as a basis to identify key molecular interacting genetic pathways in an attempt to correct these mutations and restore proper heart formation and function.

Marfan syndrome is an inherited connective tissue disorder that affects the formation and function of the heart, blood vessels, eyes and musculoskeletal elements including the joints. This debilitating syndrome has an incidence of 1:5000 and can lead to an aortic aneurysm which can be lethal if left undetected. While surgery can repair a defective aorta, which is the main blood vessel in the body that attaches to the heart and carries blood throughout the body, there is no cure for Marfan syndrome. Mutations in a gene, fibrillin-1, causes Marfan syndrome and we aim to enhance fibrillin-1 microfibril abundance by protecting them from breakdown to restore the aorta to its appropriate size to allow for proper function.
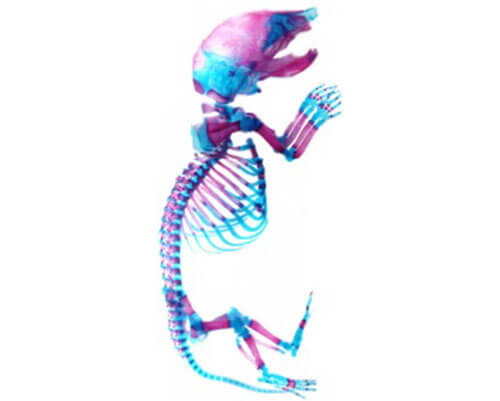
Approximately 1.5 billion people suffer from musculoskeletal disease and pain worldwide with increasing rates year after year. A subset of these conditions is a result of inherited disorders and birth defects. We have recently uncovered that mutations in a gene, ADAMTS6, results in numerous musculoskeletal malformations affecting the form and function of bones, tendons, and skeletal muscle. We aim to better understand the role of ADAMTS6 in musculoskeletal development through the use of mouse and cell culture models to uncover important molecular interactions during development to identify potential interventions.


