Targeting Plasma Cells for Organ Transplantation
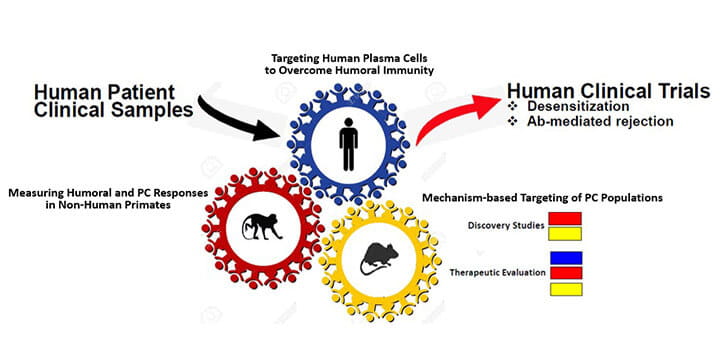 Long-lived plasma cells (PCs) are the major source of donor-specific alloantibody production, including antibodies directed against human leucocyte antigens (HLA). The aberrant production of anti-HLA antibodies presents a significant barrier to successful organ transplantation. Anti-HLA antibody production translates into difficult cross-match before transplant and increased risk of acute and chronic antibody-mediated rejection after transplant. Current evidence points towards donor- specific alloantibodies produced by PCs as the major cause of allograft loss. To mitigate allograft loss, it is essential to deplete PCs, and thereby, eliminate the source of antibodies that preclude transplantation or that promote rejection. We have successfully employed proteasome-directed PC depletion strategies in highly HLA-sensitized transplant candidates, e.g., NCT02442648.
Long-lived plasma cells (PCs) are the major source of donor-specific alloantibody production, including antibodies directed against human leucocyte antigens (HLA). The aberrant production of anti-HLA antibodies presents a significant barrier to successful organ transplantation. Anti-HLA antibody production translates into difficult cross-match before transplant and increased risk of acute and chronic antibody-mediated rejection after transplant. Current evidence points towards donor- specific alloantibodies produced by PCs as the major cause of allograft loss. To mitigate allograft loss, it is essential to deplete PCs, and thereby, eliminate the source of antibodies that preclude transplantation or that promote rejection. We have successfully employed proteasome-directed PC depletion strategies in highly HLA-sensitized transplant candidates, e.g., NCT02442648.
Genomic profiling with SC resolution represents a transformative approach to reveal the heterogeneity of PC populations in the healthy and diseased state. Single-cell RNA-sequencing methods represent a transformative means to achieve high-throughput, high-resolution transcriptomic analysis of plasma cell states and dynamics. Single-cell approaches circumvent the averaging artifacts associated with traditional bulk population data, yielding new insights into the cellular diversity underlying superficially homogeneous populations. We have used single-cell RNA-sequencing with great effectiveness to unravel the complex cell populations in healthy and diseased states and to reconstruct developmental trajectories, and modeling transcriptional dynamics. While initially PCs have been viewed mainly as short-lived end-stage B-cells, there is now evidence that this is a heterogeneous cellular compartment in which new-born plasmablasts and long-lived PCs also coexist. Our recent advances in the genomics of plasma cells with single resolution has transformed our understanding of their diversity and helped to identify novel therapeutic targets, and provide the scientific rationale for combining targeted therapies to increase tumor-cell cytotoxicity which abrogates drug resistance. A key challenge is to interpret patient profiling signatures into actionable clinical insights that make personalized therapy a practical reality. Our platforms incorporate genetic information generated from patient-derived PCs as a predictive technology that represents a revolutionary strategy that may tailor personalized medicines.


